In an age of advanced weaponry and
warfare, the risk of bioterrorism is increasingly acute. One potential
bioterrorism agent is the bacterium Francisella
tularensis, which is responsible for the similarly named disease tularemia.
F. tularensis is classified as a category
A potential bioterrorism agent, the same classification as anthrax and the
plague. A low number of bacteria are capable of causing disease, which can be
fatal in up to 60% of cases if untreated. Outside the potential threat for
bioterrorism, F. tularensis infection
also happens naturally. While cases of tularemia in the United States have
largely declined since the 1950s, this is not the case throughout the world,
with multiple outbreaks occurring in Europe in the last 10 years.
 |
| F. tularemia. CDC's Public Health Image Library. Image # 1903; photo credit: Larry Stauffer, Oregon State Public Health Laboratory. |
Disease management and bacterial
elimination can be difficult because F. tularensis can
survive in over 100 species of mammals, birds, cold-blooded animals, and
arthropods, including rabbits, mice, rats, squirrels, cats, dogs, horses, pigs,
and sheep. To further complicate matters, transmission of F. tularensis can
occur in several ways, including the consumption of contaminated water or food;
contact with urine, excrement, or blood from infected animals; bites from blood-sucking
arthropods like ticks, flies, and mosquitoes; and inhalation of aerosolized
bacteria. The symptoms of tularemia depend on the route of transmission and can
include a skin ulcer at the site of bacterial entry; swollen glands; sore
throat; and high fever. F. tularensis is
naturally resistant to many antibiotics because it is an intracellular
bacterium that spends most of its life hiding inside a host cell; an antibiotic
must first get into the host cell before it can have any effect on the pathogen.
Aminoglycosides, tetracyclines, and fluoroquinolones have been shown to be
effective, but 5-15% of infections relapse following treatment, and the side
effects from these antibiotics can be unmanageable, limiting their use.
Due to the low infectious dose,
high mortality rate, ease of transmission, and difficulty in treatment, natural F. tularensis infection is a serious threat to public
health, and a weaponized version of the bacteria could be catastrophic. To
counteract these risks, researchers have been studying how F. tularensis causes infection to identify ways to inhibit or kill
the bacteria. We know that once in the human body, F. tularensis is
taken up by phagocytic cells, such as macrophages. The job of these phagocytic
cells is to engulf the bacterium into a compartment called a phagosome for
degradation. Typically, this is how the immune system would capture and kill a
pathogen. However, in the case of F. tularensis, the
bacterium escapes from the phagosome through a process that is not well
understood to begin replicating in the cytosol of the host cell. A recent study
shed a little light on this process and found that F. tularensis is manipulating the host macrophage
in a unique way.
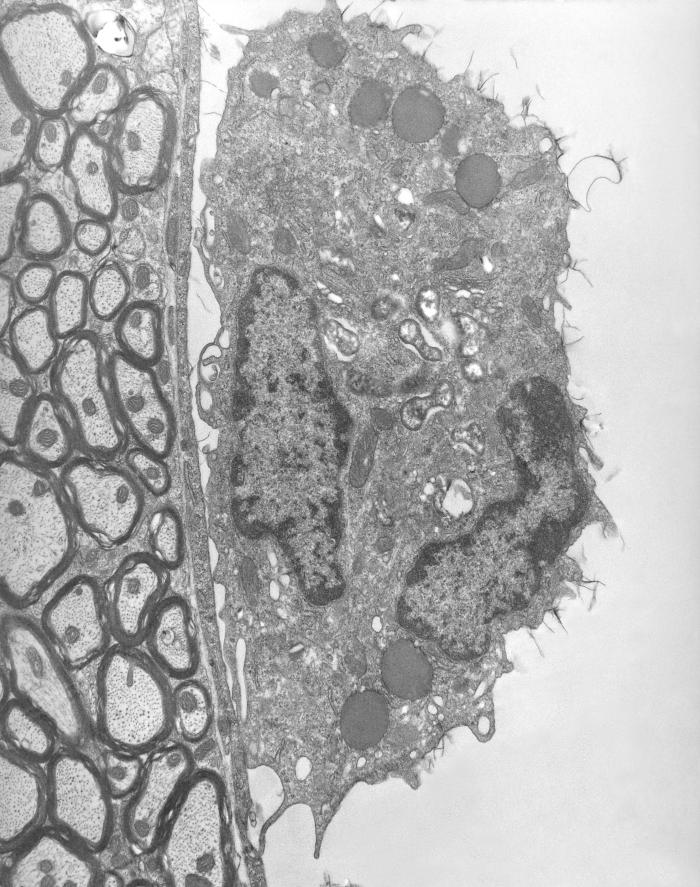 |
| Macrophage (right) containing rickettsial microbes. CDC's Public Health Image Library. Image # 8731; photo credit: CDC, Dr. Ed Ewing. |
Dr. Forrest Jessop and colleagues
at the National Institute of Allergy and Infectious Disease found that F. tularensis alters
the function of the mitochondria in the macrophage. The mitochondria are
essential cellular organelles that are responsible for providing “power” to the
cell, much like a battery provides power to a flashlight. When F. tularensis first
enters the macrophage, it improves the function of the mitochondria, which keeps
the macrophage alive and prevents an inflammatory response from the immune
system. A few hours later, the bacterium reverses these effects and decreases
mitochondrial function, decreasing the macrophage's power supply and leading to rapid bacterial replication and oncosis, a
type of cell death that involves the swelling of the cell. This facilitates the
pathogen’s ability to get out of the cell after replicating and move on to a
new host cell.
The researchers were able to take this new-found
knowledge of the bacteria’s effect on mitochondria a step further and test a
therapeutic treatment in culture. They found that by treating F. tularensis-infected
macrophages with drugs that protect typical mitochondrial function, they were
able to reduce macrophage cell death and decrease levels of bacterial replication. It
remains to be seen if this type of intervention will work in an animal system,
but this is a promising step in the right direction towards increasing the number
of treatments available for these infections. Since the environmental reservoir
for F. tularensis is so vast, increased awareness of the risks of disease and
research focus are important to stem the outbreaks and prevent future
bioterrorism threats.
No comments:
Post a Comment